Gene expression dynamics during early human brain organoid development
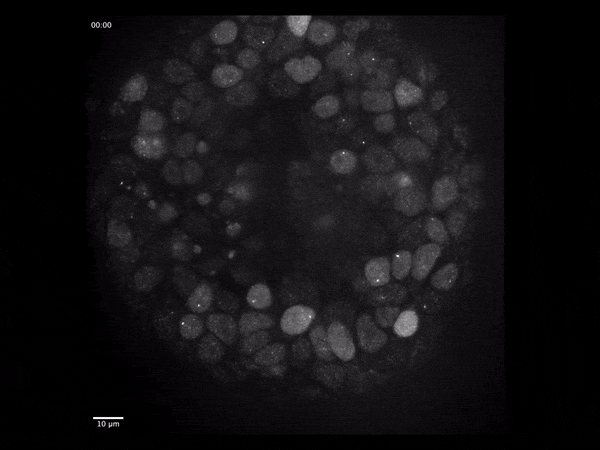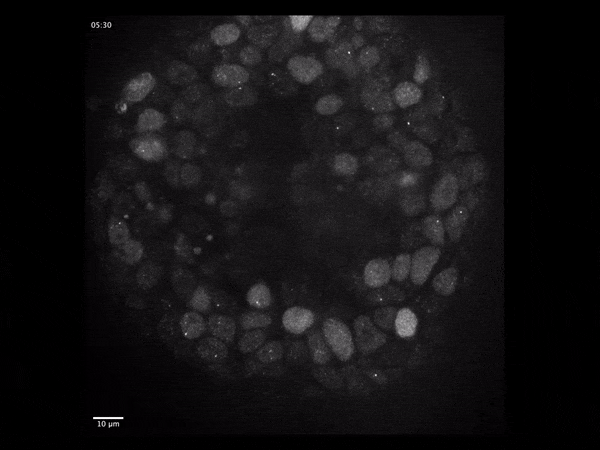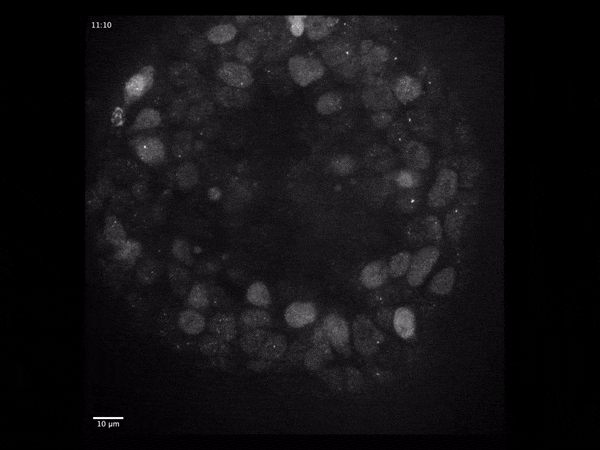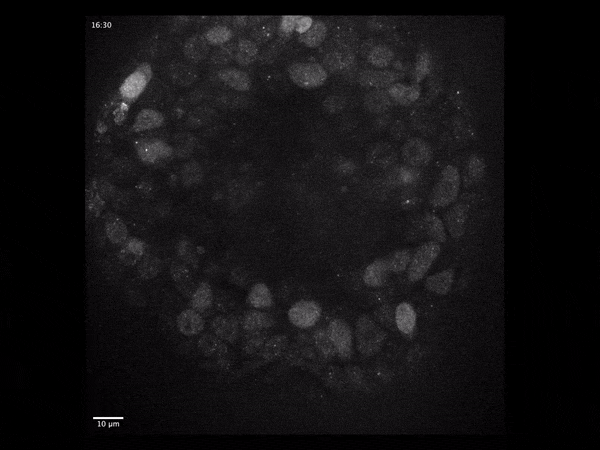
Cerebral organoids have revolutionized the study of human brain development because they model the different stages through which neural tissue transitions in vitro.
Single-cell genomics methods have provided very detailed insights into which genes are expressed in which cell types at the various stages of neurogenesis, yet little is known about how the timing and fluctuation of gene expression influences cell fate decisions and morphogenesis.
To address these questions, we have developed an imaging-based approach that allows us to monitor gene expression in real time and correlate it with cell type- and state specific information. We investigate the dynamics of gene transcription in the context of early neural development and correlate the obtained data with measurements of RNA and protein content in fixed tissues.
Ultimately, our approach will allow us to resolve how small differences in the timing, localization and frequency of gene expression result in the formation of diverse specialized cell types and tissue regions.