Research
Towards a dynamic quantitative understanding of neuronal-immune interactions in the brain
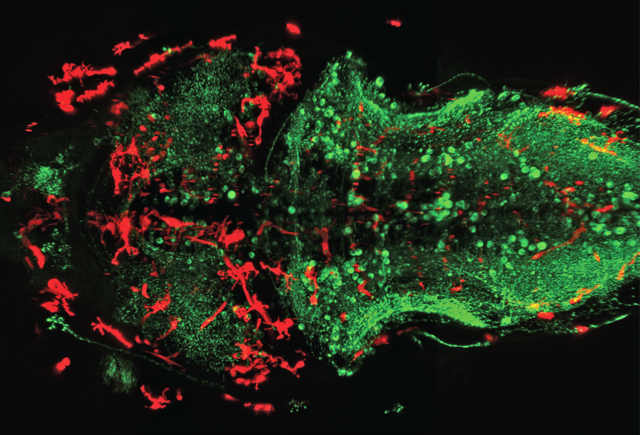
A central issue in biology is to understand how cells sense changes in the surrounding environment, integrate multiple signals and execute distinct cellular responses in vivo. We investigate this during the development of the vertebrate brain where a group of resident immune cells, the microglia, are able to identify and remove dying neurons that fail to integrate within functional circuits. Our work in the optically transparent zebrafish brain has shown that microglia form a network that surveys the brain and responds promptly to stimuli. We investigate group-level properties of this cellular network, its spatial organization, coordination and interactions with the local environment by taking a broad range of advanced fluorescence-based imaging approaches to gain a quantitative mechanistic understanding of how microglia scan their environment. By combining light-sheet microscopy for whole brain imaging with real-time signalling reporters and optogenetic methods for targeted manipulation we address how microglia identify their targets and the flipside of this, namely, how neurons communicate their status to microglia and how signals are transmitted across the brain to ensure efficient neuronal immune interactions in development and disease.
In Vivo Recording of Microglial phagocytosis
Microglia use the gastrosome, a unique subcellular compartment, to clear dying cells - The cell on the right has an expanded gastrosome.